Welcome to Significant figures & Uncertainty!
So what are significant figures you may ask?
Well...All measurements are approximations. This means that no measuring device can give a perfect measurement without an experimental uncertainty. And so, significant figures provide you with the amount of precision. For example, balance #1 can measure to the nearest thousandth of a gram, whereas balance #2 can only measure up to the nearest tenth. Therefore the results of the mass on balance #1 will have more significant figures than that of balance #2.
If you're still having trouble understanding, here are some rules to determine whether or not digits are significant:
Example:
1) 5 843 has 4 significant figures.
2) 23 has 2 significant figures.
Example:
1) 6008 has 4 significant figures.
2) 20 800384 has 8 significant figures.
Example:
1) 23.0 has 3 significant figures.
2) 740.00 has 5 significant figures.
This means that zeros to the right of a large number are not significant, and zeros to the left of a small number ( less than 1) are not either.
Example:
1) 0.0025 has 2 significant figures.
2) 65 000 has 2 significant figures.
3) 5.79 X 10 ² has 3 significant figures.
Example:
1) constants in a formula (ex. force of gravity - because it was not personally measured)
2) Values that have been counted ( ex. Bunnies)
Now, its all fine and dandy to be able to recognize significant figures, but the majority of the time, they are of no use unless you add, subtract, multiply or divide them! Heres how to do it:
The rule for addition or subtraction with significant figures:
Example:
1) 32.3 + 51 = 83.3 -> 83
(1d) + (0d)= (0d)
2) 7583.23 - 435.00023146 = 7148.22976900 -> 7148.23
(2d) - (8d) = (2d)
The rule for multiplication or division with significant figures:
Example:
1) 0.0025 x 3568 = 8.92 -> 8.9
(2sf) x (4sf) = (2sf)
2) 4525/320 = 606 -> 610 or 6.1 X 10²
(4sf)/ (3sf) = (3sf)
Well...All measurements are approximations. This means that no measuring device can give a perfect measurement without an experimental uncertainty. And so, significant figures provide you with the amount of precision. For example, balance #1 can measure to the nearest thousandth of a gram, whereas balance #2 can only measure up to the nearest tenth. Therefore the results of the mass on balance #1 will have more significant figures than that of balance #2.
If you're still having trouble understanding, here are some rules to determine whether or not digits are significant:
- Digits from 1-9 are always significant.
Example:
1) 5 843 has 4 significant figures.
2) 23 has 2 significant figures.
- Digits between two other significant digits are always significant.
Example:
1) 6008 has 4 significant figures.
2) 20 800384 has 8 significant figures.
- One or more additional zeros to the right of both the decimal and another significant digit are significant.
Example:
1) 23.0 has 3 significant figures.
2) 740.00 has 5 significant figures.
- Zeros used only for spacing the decimal point are not significant.
This means that zeros to the right of a large number are not significant, and zeros to the left of a small number ( less than 1) are not either.
Example:
1) 0.0025 has 2 significant figures.
2) 65 000 has 2 significant figures.
3) 5.79 X 10 ² has 3 significant figures.
- Numbers that are not measurements have an infinite number of significant figures, so we basically ignore them when deciding how manny significant figures we need to round to.
Example:
1) constants in a formula (ex. force of gravity - because it was not personally measured)
2) Values that have been counted ( ex. Bunnies)
Now, its all fine and dandy to be able to recognize significant figures, but the majority of the time, they are of no use unless you add, subtract, multiply or divide them! Heres how to do it:
The rule for addition or subtraction with significant figures:
- When adding or subtracting round your answer to the least number of decimal places.
Example:
1) 32.3 + 51 = 83.3 -> 83
(1d) + (0d)= (0d)
2) 7583.23 - 435.00023146 = 7148.22976900 -> 7148.23
(2d) - (8d) = (2d)
The rule for multiplication or division with significant figures:
- When multiplying or dividing with numbers round your answer to the least number of significant figures.
Example:
1) 0.0025 x 3568 = 8.92 -> 8.9
(2sf) x (4sf) = (2sf)
2) 4525/320 = 606 -> 610 or 6.1 X 10²
(4sf)/ (3sf) = (3sf)
Uncertainty (Yooopi!)
Uncertainty is present in the majority of places, this is when a quantity has been measured with an instrument. The uncertainty in the measurement is a results of the uncertainty of the instrument used or the skill of the person taking the measurement.
There are two ways to express uncertainty:
Through..
What are these foreign words you may ask? Well let me explain!
Absolute uncertainty is expressed in the same units as the measurement itself.
Example : 5.9 cm ± 0.3 cm
Relative uncertainty however is expressed as a percentage of the measuremnt. This method is easier to compare the uncertainty of more than 1 measurement.
Example: 5.9cm ± 5%
Relative uncertainty can be calculated the following way:
There are two ways to express uncertainty:
Through..
- Absolute uncertainty
- Relative uncertainty
What are these foreign words you may ask? Well let me explain!
Absolute uncertainty is expressed in the same units as the measurement itself.
Example : 5.9 cm ± 0.3 cm
Relative uncertainty however is expressed as a percentage of the measuremnt. This method is easier to compare the uncertainty of more than 1 measurement.
Example: 5.9cm ± 5%
Relative uncertainty can be calculated the following way:
Relative uncertainty = (Absolute uncertainty/ value of measurement) X 100
There are 2 main ways to determine the uncertainty of a measurement:
Example: On a balance it may say that the mass indicated has an uncertainty of 0.01g.
Example:
- Depending on the instrument sometimes the uncertainty is written on itself.
Example: On a balance it may say that the mass indicated has an uncertainty of 0.01g.
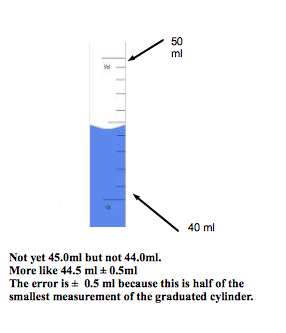
- When it is not indicated, the uncertainty is equal to one half of the smallest measurement provided by the instrument.
Example:
*Note - the uncertainty must always have the same number of decimal places and the measurement itself.
And that everybody, are significant figures and uncertainty! Hope you enjoyed.
Oh! and here are some practice sheet and a video to help you become a pro using significant figures and uncertainty.
Sig fig song:
http://www.youtube.com/watch?v=ZuVPkBb-z2I
Sig fig worksheet with answers (lucky you!):
http://www.saddleback.edu/faculty/jzoval/worksheets_tutorials/ch1worksheets/worksheet_Sig_Fig_9_11_08.pdf
Uncertainty worksheet:
http://wrean.disted.camosun.bc.ca/ph191/exercises/ph191_uncert_work.pdf
Sig fig song:
http://www.youtube.com/watch?v=ZuVPkBb-z2I
Sig fig worksheet with answers (lucky you!):
http://www.saddleback.edu/faculty/jzoval/worksheets_tutorials/ch1worksheets/worksheet_Sig_Fig_9_11_08.pdf
Uncertainty worksheet:
http://wrean.disted.camosun.bc.ca/ph191/exercises/ph191_uncert_work.pdf